  
In the oxidation number method, we consider that a complete transfer of an electron from less electronegative to more electronegative atom takes place. The more practical method is the use of oxidation number to keep track of the electron shifts happening in the chemical reactions. It is important to keep track of the electron shifts in the reactions that involve the covalent compound formations. 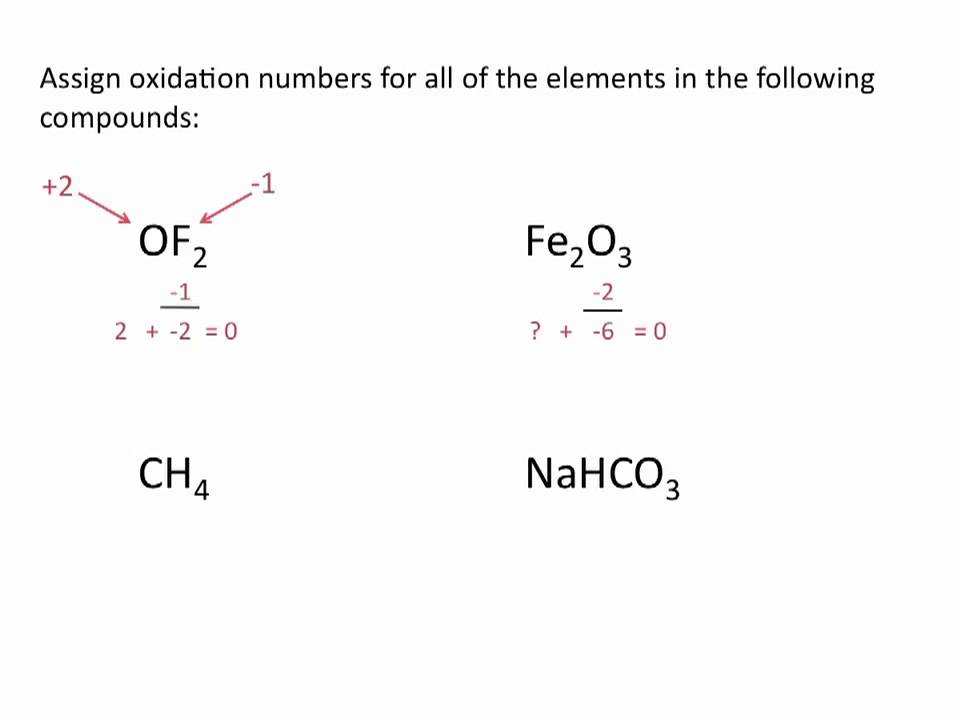
Refer to the example to understand this class of reactions: This reason is also applicable to other reaction examples as well. Therefore, it can be described as electron shift rather than electron transfer or electron loss where complete loss and gain of electrons occurs in Hydrogen and oxygen, respectively. However, when we study in depth about charge transfer we will understand that the reaction is only partial.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |




 RSS Feed
RSS Feed
